Danielle Weiss, MD; Aneela Bidiwala, MD
The Children’s Hospital at Montefiore Albert Einstein College of Medicine
Case
This is a 3-week-old female infant with trisomy 21, born at 35+2 weeks gestation with Apgar scores of 9/9 and a birth weight of 1.9 kg. At birth, she required no resuscitation and had no oxygen needs, but experienced feeding difficulties necessitating a nasogastric (NG) tube during her NICU stay. After discharge, she was asymptomatic until a sudden episode of cyanosis prompted ED evaluation. ED findings were as follows: RR 54/min, sat at 97%, normal breath sounds, no retractions. In the ED, she had desaturations to 70% which responded to HFNC 5L at 35% FiO2. There was no history of vomiting, fever, cough, or wheezing, but the mother reports fatigue and retractions during feeds.
Chest x-ray: Demonstrated left-sided consolidation and pleural effusion( Fig-1) Subsequent chest CT revealed diffuse interstitial and septal thickening (Fig 2). She was admitted for management of worsening respiratory status, requiring chest tube placement and HFNC.
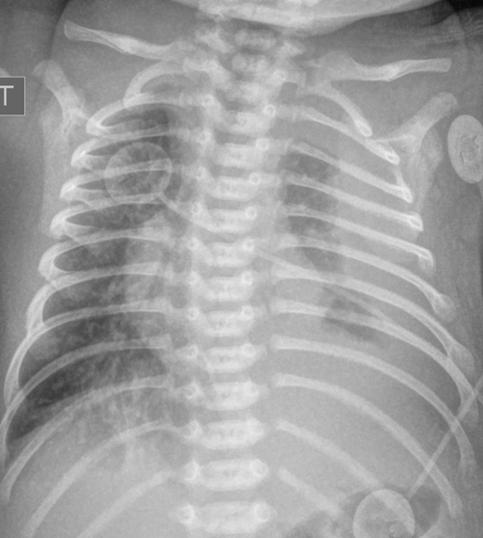
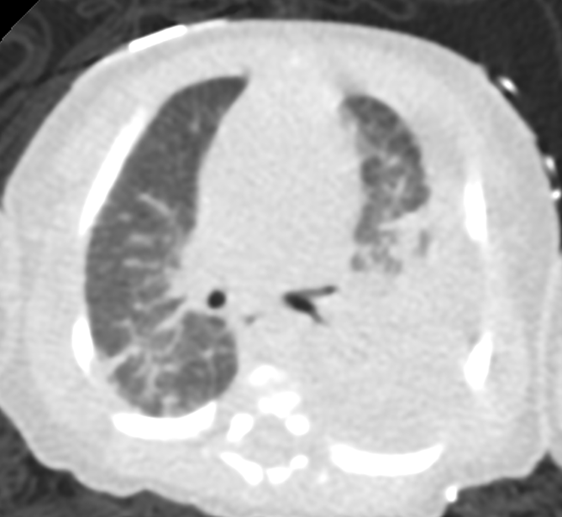
Correct!
Discussion
Our patient’s chest CT shows diffuse or segmental interlobular septal thickening, perihilar infiltrates, ground-glass opacities, and hyperinflation. These findings, when combined with pleural effusions, increase the likelihood of Congenital Pulmonary Lymphangiectasia (CPL). CPL is a rare congenital disorder of pulmonary lymphatic development. It is characterized by dilated lymphatic channels draining the interstitial and subpleural spaces of the lung. The incidence of CPL among stillborn and neonates is estimated to be <1%. The etiology of CPL is unknown, but it is suspected to have a genetic background.
Although traditionally a fatal disorder of infancy, some cases have been reported in later childhood, suggesting a spectrum of severity. Most children present in the neonatal period with respiratory distress.
Chest radiographic findings include prominent interstitial markings, hyperinflation, ground-glass opacities especially in neonates, and pleural effusion. Chest CT may show diffuse or segmental interlobular septal thickening, perihilar infiltrates, ground-glass opacities, and hyperinflation (Fig 3). Pleural fluid analysis is consistent with chylous pleural effusion. Diagnostic criteria include a milky or opalescent appearance of the pleural fluid, elevated triglyceride levels(typically >110mg/dl), and lymphocyte predominance on cell count. In neonates and young infants who may not yet be enterally fed, triglyceride levels may be lower, therefore diagnosis may rely on lymphocyte predominance(>80%) and increase in triglyceride content following initiation of enteral feeds. These findings support impaired lymphatic drainage, as seen in congenital pulmonary lymphangiectasis. Lung biopsy with histology and immunochemical staining demonstrating dilated lymphatic channels and thickening of the interlobular septa and subpleural space is the gold standard for diagnosis. Immunohistochemical staining for endothelial cell markers CD31, CD34, and D2-40 confirms lymphatic origin. Echocardiogram should be performed to rule out pulmonary venous obstruction, such as hypoplastic left heart syndrome or TAPVR, which can cause secondary lymphangiectasia. Lymphangiograms have been used in some patients to evaluate thoracic duct disruption and lymphatic duct dilation. In our patient Echocardiogram showed large left pleural effusion, PFO with bidirectional flow, mild bilateral pulmonary artery stenoses, and normal biventricular systolic function.
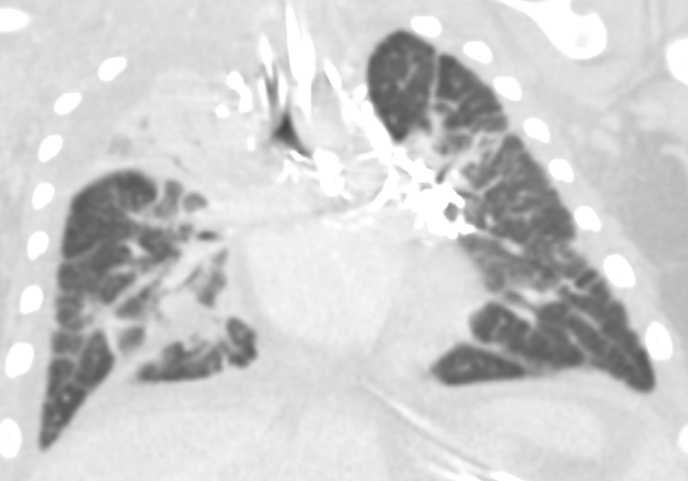
The etiology of pulmonary lymphangiectasia is unknown. Theories involve failed regression of large lymphatic channels which appear between 9 and 16 weeks gestation or bronchomediastinal lymphatic obstruction. Genes involved in lymphangiogenesis, such as vascular endothelial growth factors (VEGF-C, VEGF-D) and their receptors (VEGFR-2, VEGFR-3), have also been implicated. Prenatally, CPL may manifest as pleural effusions and hydrops. At birth, patients may have severe distress requiring mechanical ventilation. Traditionally, neonatal-onset cases were fatal. There is a well-known association with Trisomy 21, Noonan's Syndrome, and Turner Syndrome. Cases in multiple family members also suggest a genetic predisposition.
The natural course in survivors is variable. Patients presenting later in childhood tend to have more localized disease and better prognosis. Treatment, therefore, is patient-specific and mostly supportive. Apart from symptomatic and supportive treatments, novel regimens—including sirolimus (an mTOR inhibitor) and ethiodized oil lymphatic embolization under ultrasound-guided intranodal lymphangiography have shown encouraging short-term therapeutic effects for lymphatic anomalies. Surgical operations (lobectomy or pneumonectomy) can be considered for patients with CPL confined to one lobe or lung. Patients with CPL usually have a poor prognosis and often die during the neonatal period. Prognosis is expected to improve with the development of modern therapeutic agents.
References
- Yuan SM. Congenital Pulmonary Lymphangiectasia: A Disorder not only of Fetoneonates. Klin Padiatr. 2017 Jul;229(4):205-208. doi: 10.1055/s-0043-112500. Epub 2017 Jul 17.PMID: 28718185
- Reiterer F, Grossauer K, Morris N, Uhrig S, Resch B. Congenital pulmonary lymphangiectasis. Paediatr Respir Rev. 2014 Sep;15(3):275-80. doi: 10.1016/j.prrv.2014.05.002. Epub 2014 May 29.PMID: 24997116
- Adair D, Rabah R, Ladino-Torres M, Saba TG.Front. A Relatively Mild Presentation of Case Unilateral Congenital Pulmonary Lymphangiectasia. Pediatr. 2021 Apr 22;9:657473. doi: 10.3389/fped.2021.657473. eCollection 2021.PMID: 33968857
- Ladonna J Malone , Laura Z Fenton, Jason P Weinman, Miran R Anagnost, Lorna P Browne. Pediatric lymphangiectasia: an imaging spectrum.PMID: 25301383
Not quite.